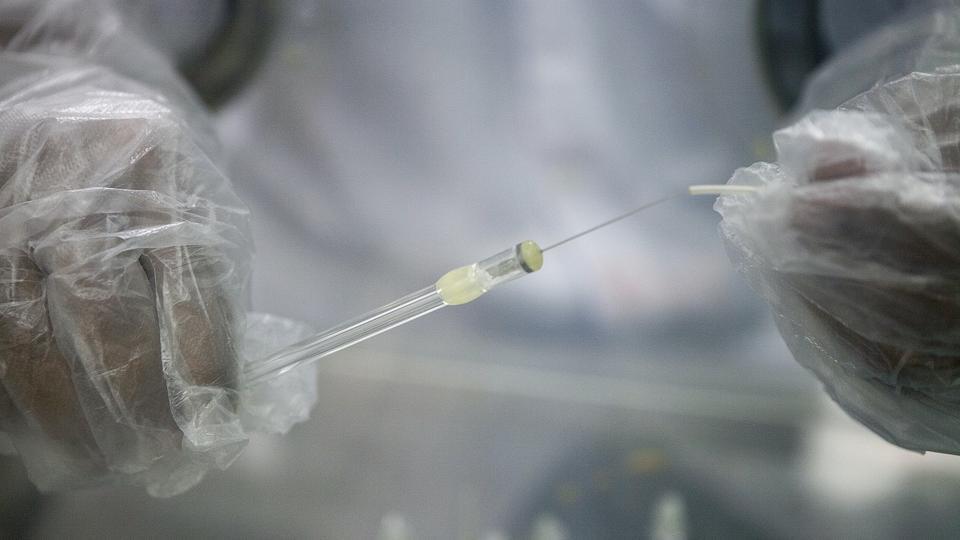
A 78-year-old Delhi-based biomedical engineer Sujoy Guha invented the drug which is a single preloaded syringe shot into the tubes carrying sperm from the testicle to the penis, under local anesthesia. The non-hormonal, long-acting contraceptive, researchers claim, will be effective for 13 years.
The clinical trials were then halted on the charge that Reversible Inhibition of Sperm Under Guidance (RISUG) was toxic. It took five years for the professor to restart the trials. In 2016, Risug is once again almost ready for its launch at the tail end of the phase III clinical trials that have been 99% successful, according to the Indian Council of Medical Research (ICMR).
After years of human trials, the drug called Risug, an acronym for reversible inhibition of sperm under guidance, is ready. It is a viscous gel which inactivates the sperm. The effectiveness of a second part of the treatment – an injection which dissolves the gel, hopefully reversing the effects and allowing a man to father a child.
Earlier this year Dr Sharma published the results of the clinical trials of the drug. Some 139 married men, under the age of 41 and having at least two children, were given a single shot of the injection and monitored for six months.
Nearly 60 years after the first birth control pill became available in the US, women today rely on more than a dozen methods of contraception to prevent unwanted pregnancies: pills and patches; injections and implants; rings and cups; and injections and sponges.
“This will be a world class contraceptive for men. It is safe and effective and lasts for long. We expect it will be cleared for production in the very near future,” says RS Sharma, a reproductive biologist at the Delhi-based Indian Council of Medical Research and the drug’s lead researcher.
Read More News On
Catch all the Health News, Breaking News Event and Latest News Updates on The BOL News
Download The BOL News App to get the Daily News Update & Follow us on Google News.